Choosing the right medical power supply is critical for ensuring the safety, reliability, and performance of medical devices. Unlike standard power adapters, medical-grade power supplies must meet strict safety standards, provide low leakage current, and deliver stable output for sensitive equipment.
Whether you are designing a patient monitoring system, diagnostic equipment, or a portable medical device, selecting the correct power solution requires careful consideration of several key factors. The power supply serves as the foundation of your medical device’s electrical architecture, directly impacting patient safety, device longevity, and regulatory compliance. Making an informed decision at the design stage can prevent costly redesigns, certification delays, and potential safety hazards in clinical environments.
Before selecting a medical power supply, make sure you have defined:
✔ Required output voltage and current
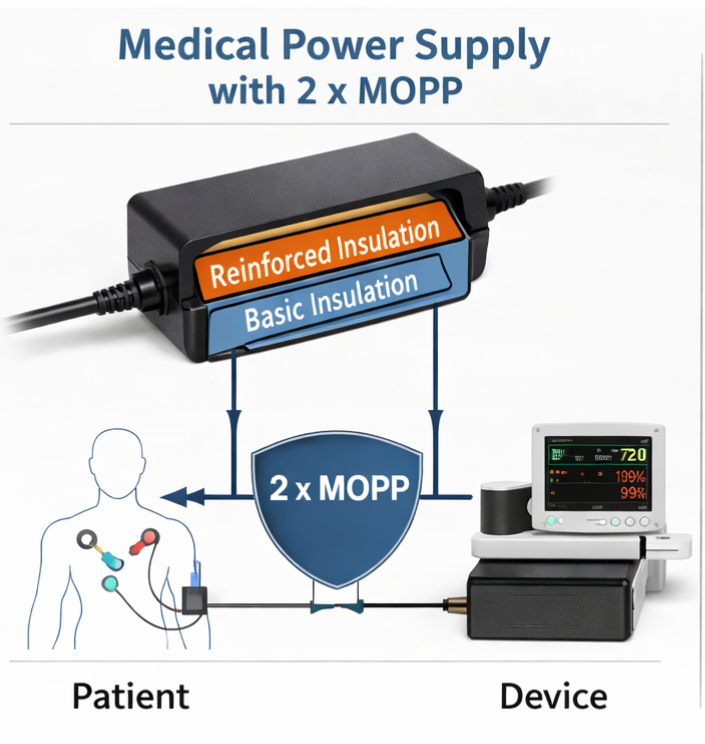
✔ IEC 60601-1 compliance level (2 x MOPP)
✔ Maximum allowable leakage current
✔ Device type (portable, stationary, patient-contact)
✔ Mechanical constraints (size, connector, cable)
✔ Certification requirements for target markets (UL, CE)
👉 This checklist helps engineers and procurement teams quickly narrow down suitable power solutions.
1. Understand Medical Safety Standards
Medical power supplies must comply with international standards such as IEC 60601-1, which defines safety requirements for medical electrical equipment. This standard is recognized globally and serves as the benchmark for medical device power supply safety.
These standards ensure:
- Low leakage current to protect patients from electrical shock
- Proper insulation (2 x MOPP – Means of Patient Protection) to prevent electrical hazards
- Patient and operator safety during normal operation and fault conditions
👉 If your device is used in direct patient contact, compliance is mandatory and non-negotiable.
Beyond IEC 60601-1, you should also be aware of regional requirements. In North America, UL 60601-1 certification is typically required. In Europe, CE marking under the Medical Device Regulation (MDR) is essential. Understanding these regulatory landscapes early in your design process will streamline certification and market entry.
More infor about IEC 60601-1 standard, please refer our: https://xjkadapter.com/iec-60601-medical-power-supply-requirements/.
2. Define Power Requirements for Your Medical Device
Incorrect power sizing is one of the most common causes of device instability and field failure.
The first technical step is to define your device’s power needs accurately. This involves calculating both the steady-state and peak power consumption of your medical device.
- Output voltage (e.g., 12V, 19V, 24V) must match your device’s input requirements
- Output current (e.g., 2A, 3A, 5A) should accommodate peak loads with adequate margin
- Total power (Wattage) determines the overall capacity needed
For example:
- Patient monitors → typically 12W–36W for continuous operation, but can reach 60W–120W for advanced systems with larger displays, multiple modules, or integrated workstations.
For example, high-end patient monitoring systems may require a 120W medical power adapter.
👉 See our case study: 19V Medical Power Adapter for Patient Monitor - Portable medical devices → 24W–65W depending on functionality
- Imaging systems → 72W–120W for high-performance applications

When determining power requirements, always include a safety margin of at least 20-30% above your calculated maximum load. This ensures the power supply operates within its optimal efficiency range and extends its operational lifespan. Additionally, consider future feature additions or software updates that might increase power consumption.
3. Consider Leakage Current
Low leakage current is one of the most important characteristics of medical power supplies and a critical safety parameter that directly affects patient protection.
A high-quality medical adapter ensures:
- Reduced risk to patients, particularly those with compromised skin integrity or implanted devices
- Compliance with IEC 60601 leakage current limits (typically less than 300μA for normal conditions)
- Stable long-term operation without degradation of insulation properties
Leakage current requirements vary depending on the type of applied part (BF, CF, or non-applied) and the intended use environment. For devices with direct cardiac contact, leakage current limits are even more stringent. When evaluating power supplies, request detailed leakage current test reports and ensure measurements were performed according to the relevant standards.
4. Evaluate Reliability and Stability
Medical devices often operate continuously in demanding environments, so power supplies must offer exceptional reliability and performance consistency.
- High efficiency (typically 85-92%) reduces heat generation and improves energy conservation
- Low ripple and noise (typically less than 150mV peak-to-peak) prevents interference with sensitive analog circuits
- Stable voltage output across varying load conditions and input voltage fluctuations
This is especially critical for sensitive equipment such as monitoring and diagnostic systems where power quality directly impacts measurement accuracy and device performance.
Consider the Mean Time Between Failures (MTBF) rating of the power supply, which indicates expected reliability. Medical-grade power supplies should offer MTBF ratings of 100,000 hours or more. Additionally, evaluate the power supply’s performance across the full operating temperature range your device will encounter in clinical settings.
5. Choose the Right Form Factor
Depending on your device architecture and application environment, you may need different power supply configurations:
- Desktop adapters for stationary equipment requiring higher power levels
- Wall plug adapters for space-constrained applications and consumer-friendly designs
- Open frame power supplies for integration into larger medical systems
Portable devices require compact and lightweight designs that don’t compromise on safety or performance, while stationary equipment may prioritize stability and power capacity over size constraints.
The physical design should also consider cable management, connector durability, and ease of replacement in clinical settings. Medical environments demand robust mechanical design that can withstand frequent cleaning, accidental impacts, and the rigors of daily clinical use.
6. Consider Customization (OEM/ODM)
Many medical devices require customized power solutions tailored to specific application requirements, including:
- Special connectors that meet your device’s mechanical and electrical interface requirements
- Cable length adjustments to optimize usability in clinical workflows
- Custom enclosure design for seamless integration with your device aesthetics
- Certification support to navigate complex regulatory pathways efficiently
Working with an experienced manufacturer ensures your power supply meets both technical and regulatory requirements while optimizing for cost, performance, and time-to-market.
A capable OEM/ODM partner can provide valuable design guidance, prototype development, regulatory expertise, and scalable manufacturing capabilities. They should demonstrate experience with medical device certifications and maintain quality management systems compliant with ISO 13485, the international standard for medical device quality management.
Choosing a reliable medical power supply manufacturer is critical.
Look for suppliers that:
✔ Have IEC 60601-1 certified products
✔ Support OEM/ODM customization
✔ Provide complete test reports
✔ Have experience with global certifications (UL, CE, FCC)
✔ Offer stable mass production capability
Conclusion
Selecting the right medical power supply is not just about matching voltage and current specifications. It involves comprehensive evaluation of compliance with safety standards, ensuring low leakage current for patient protection, and designing for long-term reliability in demanding clinical environments.
The decision requires balancing technical performance, regulatory compliance, cost considerations, and manufacturing scalability. By carefully evaluating each of these factors and partnering with experienced suppliers, you can ensure your medical device has a power supply foundation that supports both patient safety and commercial success.
👉 For a complete overview of medical power solutions, visit our Medical Power Supply Solutions page.
✅ FAQ – Medical Power Supply Selection
1. What is IEC 60601-1 and why is it important for medical power supplies?
IEC 60601-1 is the international safety standard for medical electrical equipment. It defines requirements for electrical safety, insulation, and leakage current to ensure patient and operator protection.
For any device used in healthcare environments, especially those with patient contact, using an IEC 60601-1 compliant power supply is mandatory for certification and market approval.
2. What is 2 x MOPP in a medical power supply?
2 x MOPP (Means of Patient Protection) refers to a high level of electrical isolation designed to protect patients from electric shock.
It ensures:
- Double insulation between input and output
- Safe operation even under fault conditions
- Compliance with medical safety standards
Medical devices with direct patient contact typically require 2 x MOPP certified power supplies.
3. What is acceptable leakage current for medical power supplies?
Leakage current limits depend on the application, but typically:
- General medical devices: less than 300 μA
- Patient-connected devices (BF/CF type): significantly lower limits
Lower leakage current reduces the risk of electrical shock and is critical for patient safety, especially in sensitive applications.
4. Can I use a standard power adapter for medical devices?
No, standard power adapters are not suitable for medical devices.
Medical power supplies are specifically designed to:
- Meet IEC 60601-1 safety standards
- Provide low leakage current
- Ensure proper isolation (2 x MOPP)
Using non-medical adapters can lead to certification failure and safety risks.
5. How do I choose the right power rating for my medical device?
You should consider:
- Output voltage required by the device
- Maximum current consumption
- Peak load conditions
It is recommended to select a power supply with at least 20–30% margin above your device’s maximum power requirement to ensure stability and long-term reliability.
6. What is the difference between medical and industrial power supplies?
Key differences include:
- Medical power supplies comply with IEC 60601-1
- Much lower leakage current requirements
- Higher isolation levels (2 x MOPP)
- Designed for patient safety
Industrial power supplies do not meet these strict medical safety requirements.
7. Do I need UL or CE certification for medical power supplies?
Yes, depending on your target market:
- North America → UL 60601-1
- Europe → CE marking (MDR)
- Global markets → IEC 60601-1 baseline
Choosing a pre-certified power supply can significantly reduce your product certification time.
8. When should I consider a custom (OEM/ODM) medical power supply?
You should consider customization if your device requires:
- Special connectors
- Unique voltage/current specifications
- Specific cable lengths
- Integration into a compact enclosure
Working with an experienced OEM manufacturer ensures compliance, faster development, and better system integration.
9. What form factor is best for my medical device?
It depends on your application:
- Desktop adapters → high power, stationary equipment
- Wall plug adapters → compact, portable devices
- Open frame → integrated systems
Selecting the right form factor improves usability and system design efficiency.
10. How do I choose a reliable medical power supply manufacturer?
Look for suppliers that:
- Offer IEC 60601-1 certified products
- Have experience in medical applications
- Provide full test reports
- Support OEM/ODM customization
- Ensure stable mass production
A reliable manufacturer helps reduce risks in certification, production, and long-term supply.




