Introduction
Medical power supplies operate in critical environments where human safety is paramount, making strict adherence to international safety standards absolutely essential. These standards exist to ensure comprehensive protection for both patients receiving treatment and healthcare operators working with medical equipment. Among the various regulatory frameworks governing medical electrical equipment, IEC 60601-1 stands as the most critical and widely recognized standard worldwide.
For engineers designing medical devices and procurement professionals selecting components, a thorough understanding of this standard is not merely beneficial—it is absolutely essential when evaluating, selecting, or designing a medical power supply for your specific device application.
What is IEC 60601-1?
IEC 60601-1 represents the cornerstone international safety standard that establishes comprehensive basic safety and essential performance requirements for medical electrical equipment and medical electrical systems.
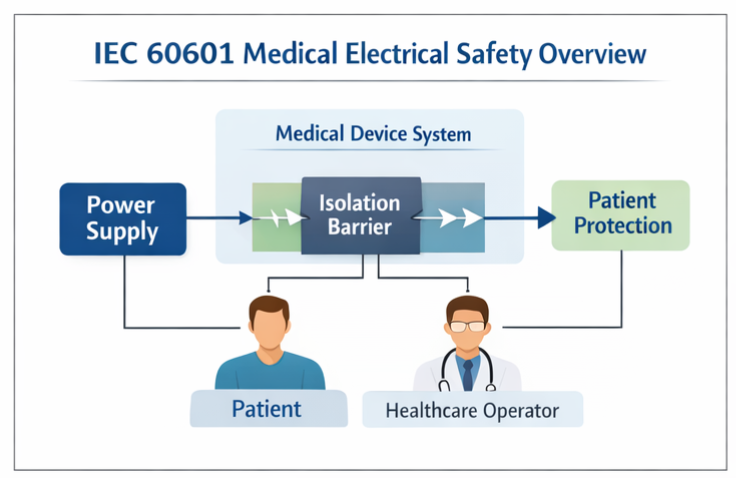
This standard focuses on three fundamental safety pillars:
- Electrical safety to prevent hazardous voltages and currents
- Mechanical safety to protect against physical hazards
- Protection against electric shock for both patients and operators
The standard’s scope is intentionally broad, applying to medical devices deployed across hospitals, outpatient clinics, specialized medical facilities, and increasingly important home healthcare environments where professional supervision may be limited.
Key Requirements for Medical Power Supplies
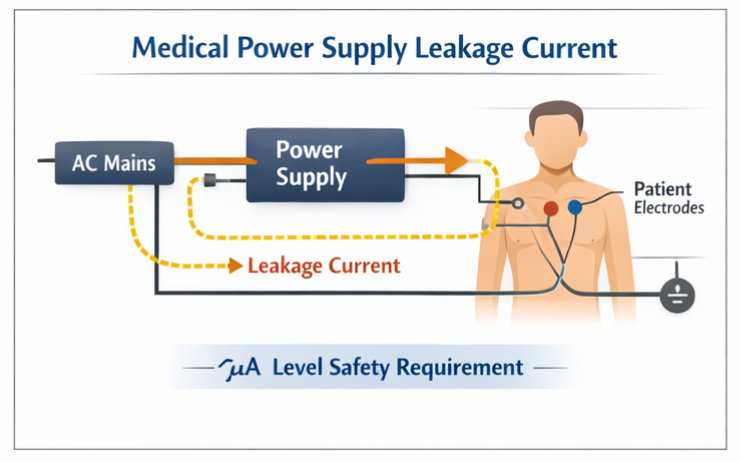
🔹 Low Leakage Current
Medical power supplies must strictly limit leakage current to extremely low levels—often measured in microamperes—to ensure absolute patient safety. This requirement becomes particularly critical for equipment that maintains direct physical contact with patients, where even minimal leakage currents could pose serious risks. The standard defines specific maximum leakage current thresholds for different equipment classifications, with the most stringent limits applied to patient-connected devices that could create a direct conduction path to the heart or other vital organs.
🔹 MOPP (Means of Patient Protection)
IEC 60601 establishes specific and rigorous insulation level requirements based on the equipment’s intended use and patient contact classification:
- 2 x MOPP (two independent Means of Patient Protection) for equipment with applied parts that directly contact patients
- Reinforced insulation barriers between primary input circuits and secondary output circuits to ensure multiple layers of protection
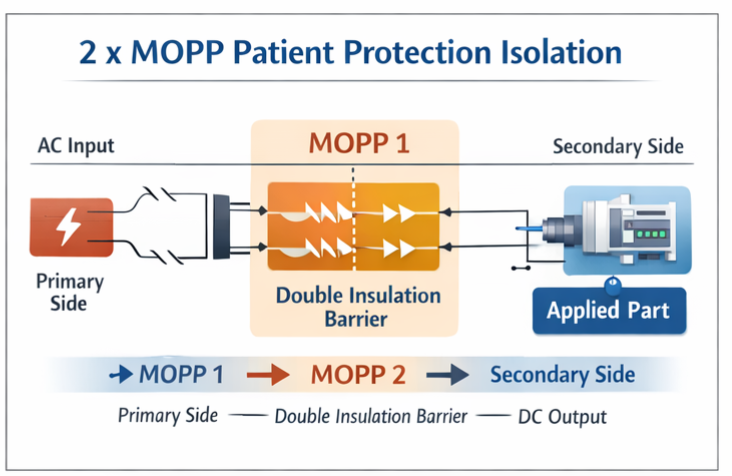
These requirements ensure that even in the event of a single insulation failure, patients remain protected from hazardous voltages, providing redundant safety mechanisms that are fundamental to medical device design philosophy.
🔹 Isolation Voltage
Medical power adapters and supplies must provide exceptionally high isolation voltage—typically 4kV (4000 volts) or higher depending on the specific application and equipment classification—to effectively prevent electrical hazards. This high isolation voltage creates a robust barrier between potentially hazardous mains voltage and patient-accessible circuits, ensuring that fault conditions cannot compromise patient safety. The isolation voltage requirement is verified through rigorous high-potential (hipot) testing during both design validation and manufacturing quality control processes.
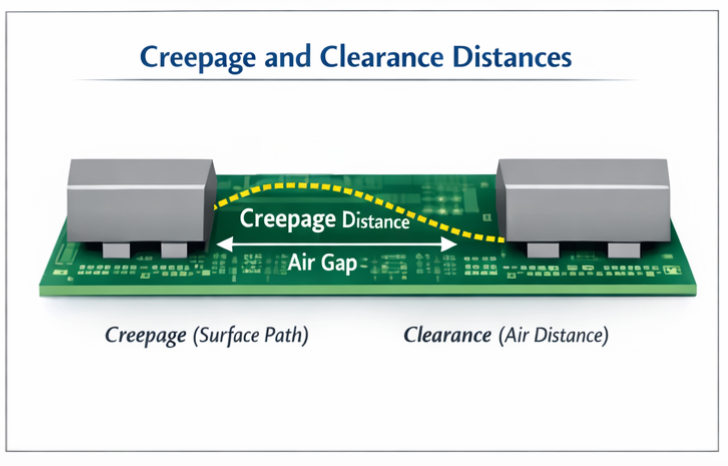
🔹 Creepage & Clearance
The physical design of medical power supplies must maintain carefully calculated safe distances between conductive parts to prevent electrical breakdown, arcing, or tracking across insulating materials. Creepage distance refers to the shortest path along the surface of insulating material between conductive parts, while clearance represents the shortest direct air path between conductors. These distances are determined by factors including working voltage, pollution degree of the operating environment, and material group of the insulation. Proper creepage and clearance design prevents both immediate breakdown and long-term degradation that could compromise safety.
IEC 60601 vs IEC 62368
While IEC 62368 serves as the applicable safety standard for information technology equipment, audio/video equipment, and communication technology equipment, it fundamentally does not meet the significantly more stringent safety requirements specifically needed for medical device applications where patient safety is directly at stake.
| Feature | IEC 60601 | IEC 62368 |
|---|---|---|
| Application | Medical | IT |
| Leakage current | Very low | Standard |
| MOPP | Required | Not required |
👉 Therefore, medical applications must exclusively use IEC 60601-certified power supplies rather than general-purpose power supplies certified only to IT equipment standards, regardless of apparent technical similarities.
Why Compliance Matters
Utilizing a fully compliant medical power supply that meets all applicable IEC 60601 requirements ensures multiple critical benefits:
- Patient safety through proven protective measures and redundant safety mechanisms
- Regulatory approval from medical device authorities including FDA, CE marking bodies, and other international regulators
- Reduced liability risks and legal exposure for manufacturers and healthcare facilities
- Market access across international jurisdictions with varying regulatory requirements
- Professional credibility and trust with healthcare providers and patients
Non-compliance can result in certification delays, costly redesigns, market access denial, and most importantly, potential patient harm.
How to Choose the Right IEC 60601 Power Supply
While IEC 60601 defines the safety requirements for medical power supplies, selecting the right model also depends on your specific application, including output voltage, power level, and patient safety classification.
For a practical step-by-step guide on selecting the correct medical power supply for your device, including voltage selection, MOPP level, and application scenarios, please refer to our detailed guide below: How to Choose a Medical Power Supply for Your Application.
This guide is designed for engineers and procurement teams working on:
- Patient monitor power supply systems
- Medical workstations
- Portable diagnostic devices
It provides a structured selection framework to help ensure both compliance and system reliability.
Conclusion
IEC 60601-1 compliance is not an optional consideration or a competitive differentiator for medical devices—it represents a fundamental, non-negotiable requirement for any equipment intended for medical use. Choosing a properly certified and compliant power supply is absolutely critical for ensuring patient and operator safety, achieving device reliability under demanding medical conditions, and obtaining successful regulatory certification
Looking for a reliable IEC 60601-compliant power adapter?
Explore our medical power supply solutions for patient monitoring, portable workstations, and imaging systems.
❓ FAQ: IEC 60601 Medical Power Supply Standards
What is an IEC 60601 power supply?
An IEC 60601 power supply is a medical-grade power supply designed to meet strict safety and performance requirements defined by the IEC 60601-1 standard. It ensures safe operation in medical environments, especially for devices that come into contact with patients.
What does 2 x MOPP mean in medical power supplies?
2 x MOPP (Means of Patient Protection) refers to two independent layers of insulation that protect patients from electric shock. It is required for medical devices with direct patient contact, such as patient monitoring systems and ECG equipment.
Why is leakage current important in medical power supplies?
Leakage current must be extremely low in medical applications to prevent electrical hazards. For patient-connected devices, leakage current is typically limited to microampere (µA) levels to ensure patient safety and regulatory compliance.
What is the difference between IEC 60601 and IEC 62368 power supplies?
IEC 60601 applies to medical electrical equipment and includes strict requirements for patient safety, such as MOPP and low leakage current. IEC 62368 is used for IT and AV equipment and does not meet the same level of safety required for medical devices.
What type of power supply is used for patient monitoring systems?
Patient monitoring systems typically require a medical power supply with 2 x MOPP, low leakage current, and stable output—commonly a 19V medical power adapter depending on the system design.
Do all medical devices require IEC 60601 power supplies?
Not all devices require the same level of protection, but any equipment used in medical environments—especially those with patient contact—must comply with IEC 60601 standards to ensure safety and regulatory approval.



